
Celebrating World Sight Day with 5 Extraordinary Ophthalmic Companies
“Who are the most innovative companies in ophthalmology?”
This simple question got an amazing response on LinkedIn with over 30 companies being recommended to me.
Having spoken to many ophthalmic professionals across the pharmaceutical, diagnostic and surgical spaces, I’ve learned about countless revolutionary concepts that will improve outcomes for patients across the globe.
We could spend all day talking about the innovations of ophthalmic giants like Alcon, J&J, Bausch & Lomb and Carl Zeiss, however, with their huge R&D budgets, innovation should be expected right?
Here are five growing companies that you might not know about who could change the future of the industry:
GlobeChek

In the US, over 40 million people have some form of vision loss, with 50% undiagnosed. GlobeChek are combating this issue, bringing diagnosis to point of care (PoC).
Founded by doctors William J. Mallon and Adam M. Katz, this company is the first to offer a comprehensive tele-ophthalmology solution in the form of a mobile kiosk. Their globe-like device provides a full diagnostic portfolio, thanks to partnerships with market leading diagnostic companies – Topcon, Visionix and Essilor.
GlobeChek screening examination includes fundus photography, external photography, visual acuity distance & near, auto refraction, eye pressure, OCT macula, OCT optic nerve, angle measurement' corneal thickness, corneal topography and cataract screening.
These examinations are carried out via the device in ‘pop-up’ locations with heavy foot traffic like schools, shopping centres and airports, offering diagnosis at PoC.
With a technician on hand to walk patients through the process, these diagnoses are made easily thanks to the use of their proprietary GlobeChek Reading Center. Patients are then provided with advice on the best course of action to take.
What all this means is more patients can be diagnosed more quickly, potentially making standard practices a thing of the past.
This device has further potential to be integrated with AI, meaning even faster and more accurate diagnosis. GlobeChek is currently selling its ESG 1200 model, which has had a positive response.
Avellino Labs
Avellino Labs are also entering the commercialisation stage of their business strategy.
Innovating in diagnostics and pioneering in the genetics space, they’ve introduced the world's first DNA/gene testing eye exam for corneal diseases.
Bringing genetic testing into mainstream ophthalmology means that we’ll be able to look at a patient’s DNA to diagnose hereditary diseases.
This proactive approach to diagnostics means that patients could be treated or monitored before their disease develops and becomes an issue. This also means that family members of diagnosed patients can be tested to catch diseases early.
The Avellino Labs test is non-invasive and performed with a quick, buccal swab of the mouth, meaning minimal discomfort for the patient. The DNA is then extracted from the swab, sequenced and analyzed for over 1,000 genetic indicators, across 75 genes associated with keratoconus and 70 mutations associated with TGFBI corneal dystrophies.
Currently, test results take up to two weeks. However, Eric Bernabei, Avelino’s Chief Sales and Marketing Officer, informed me that the ultimate goal is to get this information to the physician within as little as two days.
Since this test looks for variants and risk factors, there is no FDA approval currently required. However, it is fully CLIA validated in the US meaning that it is fully compliant to laboratory standards and commercialisation is now underway.
Preceyes
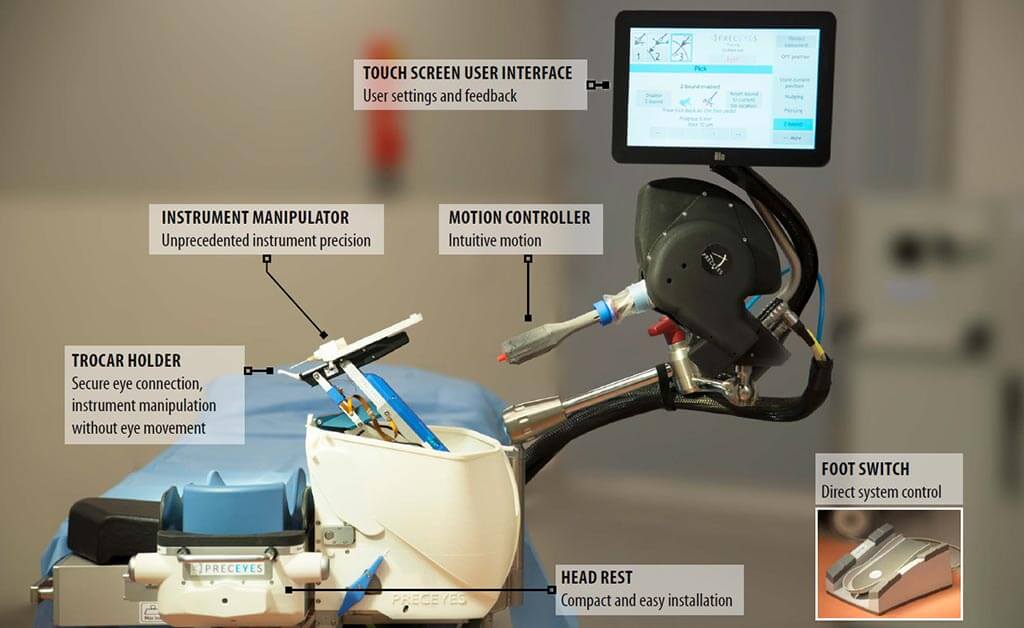
Surgical robotics company, Preceyes, are also undergoing a major milestone having just received CE marking for their surgical system. This device is the first ever market-cleared robotic assisted system to be used in vitreoretinal surgery.
Their system holds instruments steady to 20 micrometres, eradicating the issue of fatigue, meaning more precise and safer procedures. This is a crucial quality within the intricate world of retinal surgery.
Although not by any means fully automated, the system can also record movements. This means that the more the devise is used, the greater amount of data-based approaches and objective decision making can be incorporated into the surgical procedure. These advantages extend to outside the surgical process, helping evaluate surgery and for training purposes.
Moving forward, Chief Commercial Officer Preceyes, Dr Kester Nahen, explained that advanced visualisation and imaging will help guide and automate surgical steps.
While the product is now ready for commercialisation, production is not quite there yet. It’s likely that an increase in external funding will see the product’s market entry in 2020.
Due to its delicate relationship with surgical procedures, training will also need to be provided to help surgeons get used to their new robo-assistant.
Keranova

While Preceyes are innovating in retinal surgery, Keranova are disrupting anterior surgery.
Cataract surgery has had little innovation in its recent history, with phacoemulsification invented more than 40 years ago, a manual technique that’s still used in 90% of surgeries today.
However, Keranova are changing the process by performing a photoemulsification, combining a robotic arm, a next-generation ultra-fast femto-second laser and an irrigation/aspiration module with phacoemulsification capabilities.
Their CEO Fabrice Romano, who has over 30 year of surgical experience, explained to me that his company’s mission is to make surgery more accurate and efficient.
Understanding the difficulties that surgeons face, their all-in-one device can make incisions, capsulotomies and emulsify the cataracts in just 30 seconds. Providing the surgeon with the integrated irrigation/aspiration module, additionally it allows the surgeon to remove debris, saving time and additional expenses.
The intraocular lense does still need to be inserted by a surgeon. However, the automation of the femto/phaco aspect of surgery makes the process quicker and safer. All this means that more patients can be treated for this increasingly common procedure.
Having recently raised more than $30m since its inception three years ago, Keranova will now move onto their second round of clinical trials and the manufacturing of more prototypes.
We’re likely to see this device brought to market by 2022.
Medicom Healthcare
When you think of innovation, you’ll often picture these high-tech robotic systems and cool gadgets first – maybe that’s why I’ve left this pharmaceutical dealing in generics until last.
Nevertheless, Medicom Healthcare are doing things their own way and taking the market by storm.
They’ve been around over a decade, but things are really kicking off for them now with their branded range, Evolve.
Growing rapidly, they’re the biggest supplier of ophthalmic generic medicines to the NHS and supply over 25 countries globally. Their dry eye brand, Evolve, is now the fastest growing product range for dry eyes in the NHS, achieving market leadership in over 10 CG localities in the UK in just over two years.
Head of Europe, Tom Harrington, explained that Medicom’s mission is to offer quality and affordable preservative free products for everyone. Their launch portfolio is also planned to offer new preservative free medicines in therapy areas addressing major problems in eye health like glaucoma, allergy and infection.
Their current Evolve products offer a range of excellent preservative free formulations covering, at competitive prices both for the NHS and OTC markets. With the patient and consumer at the heart of what they do, the Evolve range has proven to be attractive to both consumers and payers.
There you have it. Five extraordinary companies disrupting ophthalmology in their own way. As they receive approvals, become commercialised and grow, they’re sure to impact ophthalmology in a big way – with other big corporations in this space taking notice.
What’s next? I can’t wait to see.
If you’d like to learn more about these exciting companies or would like to tell me about your company, please email me at Becky.Rowlands@medical-cm.com.
For more content like this visit my consultant page.
Recommended.
Breakthroughs in Modern Cataract Surgery.
In 2023, what advancements can be seen across cataract surgery and the wider ophthalmology space? I’ve explored some of the more impressive new tech.

What Does AI Really Mean For Ophthalmology?
AI advancements are leading to huge developments in ophthalmology. What does this mean for clinicians and patients? Click to find out my thoughts.

Glaucoma Companies with 2022 Vision.
I’m always interested in companies focused on innovative devices and products, so was intrigued by these players in the glaucoma market. Click here to find out who.
.jpg)
5 Innovative Surgical and Diagnostic Products Changing the Industry Landscape.
As frequency of eye disease rises with aging populations, these industry innovators are enabling earlier disease detection and creating more efficiency in the process. Click to find out more.
Comments.